New discovery of fouling mechanism of nanofiltration membrane
According to the WeChat public platform of Membrane Science and Engineering on April 19. 2019. although membrane separation technology has advantages such as green and high efficiency, in practical applications, solutes will inevitably accumulate/adsorb on the membrane surface or pores, forming membrane fouling. Traditional theory suggests that membrane fouling, such as fouling of filter cake layers or pores, can lead to an increase in mass transfer resistance of the membrane, resulting in an increase in solute retention rate under constant flux conditions. However, recently, when studying the pollution mechanism of polycyclic aromatic hydrocarbons (PAHs) in water on polyamide nanofiltration membrane, Wan Yinhua, a researcher team from the Institute of Process Engineering, Chinese Academy of Sciences, found that although membrane pollution would lead to a decrease in the permeation flux of nanofiltration membrane, its rejection rate of neutral hydrophilic small molecules under constant flux operation remained unchanged.
PAHs are organic micro pollutants composed of multiple benzene rings, with strong teratogenicity and carcinogenicity, mainly derived from incomplete combustion of various carbon materials such as fossil fuels and biomass. Various types of industrial wastewater (mainly coking wastewater) contain a large amount of PAHs. Due to the high efficiency of nanofiltration membranes in intercepting small molecule organic compounds, they are usually used as the final step in wastewater treatment. Therefore, analyzing the pollution mechanism of PAHs on nanofiltration membranes has important guiding significance for the treatment of wastewater containing PAHs. The PAHs present in coking wastewater mainly include naphthalene and its methylated derivatives, as well as PAHs composed of three to five benzene rings, with molecular weights generally below 500 Da. Based on this, researchers selected five commercial polyamide nanofiltration membranes to study their interception and pollution mechanisms in the treatment of simulated and real coking wastewater containing PAHs. Naphthalene, 6-methoxy-1.2.3.4-tetrahydronaphthalene, and pyrene (containing four benzene rings) were selected as model pollutants.
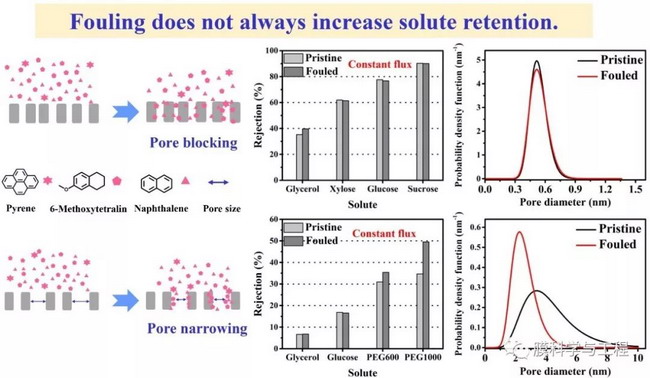
The research team found that polyamide nanofiltration membranes have strong adsorption capacity for PAHs. In the early stage of filtration, the adsorption of PAHs on the nanofiltration membrane is the main retention mechanism. However, due to the dissolution diffusion effect, the PAHs adsorbed by the membrane will gradually diffuse and desorb through the membrane, resulting in a decrease in their apparent retention rate. Molecular exclusion is another important mechanism for the removal of PAHs by nanofiltration membranes. The larger the molecular weight of PAHs, the weaker their polarity, and the lower their adsorption on the membrane, the stronger the membrane's exclusion effect on their molecules. However, although the adsorption of PAHs can cause a decrease in membrane permeate flux and porosity, it does not necessarily lead to a decrease in the average pore size of the nanofiltration membrane and an increase in the retention rate of neutral small molecules. By analyzing the pore size distribution before and after treating PAHs with nanofiltration membranes, it was found that for membranes with smaller pore size and narrower pore size distribution (such as NF270), PAHs will adsorb indiscriminately in almost all membrane pores. Although this indiscriminate adsorption can cause membrane pore blockage, the pore size distribution and average pore size of the membrane remain basically unchanged. For membranes with larger pore size and wider pore size distribution (such as NF10), PAHs are mainly adsorbed in the large pores of the nanofiltration membrane (low diffusion resistance, high local flux), resulting in a shrinkage effect that narrows the pore size distribution and reduces the average pore size. However, due to the fact that the pore size of the nanofiltration membrane after adsorbing PAHs is still much larger than the hydration radius of some hydrophilic neutral small molecules (such as glucose and glycerol), the adsorption of PAHs will not significantly affect the membrane's ability to retain them.
When using nanofiltration membranes to treat real coking wastewater containing PAHs, the inorganic salts and other organic compounds contained in the wastewater can cause more severe membrane fouling, resulting in a significant decrease in membrane flux. However, the retention rate of PAHs slightly increases when the membrane fouling reaches a certain level. By simple chemical cleaning, PAHs adsorbed on/inside the membrane can be basically removed, but PAHs in the cleaning solution can still cause secondary pollution.
The research was funded by the Major Science and Technology Program for Water Pollution Control and Treatment (2017ZX07402001-004) and the Youth Innovation Promotion Program of the Chinese Academy of Sciences (2017069). The relevant research results were published in Journal of Membrane Science, 582. 2019. 264-273.






